What is long term potentiation in psychology? This question unlocks a fascinating exploration into the very foundations of how our brains learn and remember. Prepare to delve into the intricate dance of neurons, where strengthened connections pave the way for enduring memories, a process crucial for understanding cognition.
Long-term potentiation, or LTP, represents a persistent strengthening of synapses based on recent patterns of activity. It’s a fundamental mechanism believed to underlie learning and memory in the brain. This process involves changes at the cellular and molecular level, enhancing communication between neurons and solidifying information for later retrieval. From its initial discovery to its complex mechanisms and widespread influence across various brain regions, LTP offers profound insights into the dynamic nature of our neural circuitry.
Foundational Concepts of Long-Term Potentiation (LTP)

Long-term potentiation, or LTP, is a cornerstone concept in understanding how our brains learn and remember. At its heart, LTP represents a persistent strengthening of synapses, the junctions between neurons, based on recent patterns of activity. Think of it as a biological form of “practice makes perfect” for neural pathways. This enduring increase in synaptic efficacy is widely considered a primary cellular mechanism underlying learning and memory formation.The intricate dance of LTP involves a cascade of cellular and molecular events.
When a neuron is repeatedly stimulated, its connections with other neurons become more robust. This isn’t a fleeting change; it’s a long-lasting enhancement that can persist for hours, days, or even longer. The key players in this process include neurotransmitters, receptors, and various signaling molecules that work in concert to modify the strength of synaptic transmission.
Definition and Significance of Long-Term Potentiation
Long-term potentiation is fundamentally defined as a long-lasting increase in synaptic strength between two neurons that results from synchronous, high-frequency stimulation. This potentiation is expressed as an enhanced postsynaptic response to presynaptic stimulation, meaning the receiving neuron becomes more likely to fire after the sending neuron releases its neurotransmitters. The significance of LTP lies in its pervasive role in various forms of learning and memory, from simple associations to complex cognitive tasks.
It provides a tangible, observable change in neural circuitry that can be directly linked to behavioral modifications associated with learning.
Cellular and Molecular Mechanisms of LTP
The molecular underpinnings of LTP are complex and have been extensively studied, particularly in the hippocampus, a brain region crucial for memory. A key pathway involves the N-methyl-D-aspartate (NMDA) receptor, a type of glutamate receptor. For LTP to be induced, glutamate must bind to both NMDA and AMPA receptors, and the postsynaptic membrane must be sufficiently depolarized. This depolarization relieves a magnesium ion block on the NMDA receptor, allowing calcium ions to flow into the postsynaptic neuron.
The influx of calcium triggers a series of intracellular signaling cascades.These cascades can lead to several outcomes that strengthen the synapse:
- Increased insertion of AMPA receptors into the postsynaptic membrane, making it more sensitive to glutamate.
- Phosphorylation of existing AMPA receptors, enhancing their conductance.
- Structural changes, such as an increase in the size of the postsynaptic density, the region where receptors are concentrated.
- In some cases, retrograde signaling molecules are released from the postsynaptic neuron, which can affect presynaptic neurotransmitter release, further enhancing transmission.
The enduring nature of LTP is thought to involve changes in gene expression and protein synthesis, leading to more permanent alterations in synaptic structure and function.
Historical Context of LTP Discovery
The initial observations that hinted at LTP’s existence emerged in the early 1970s. Researchers were investigating synaptic transmission in the hippocampus of rabbits. During experiments designed to study how neurons respond to electrical stimulation, they noticed that a brief period of high-frequency stimulation led to a lasting increase in the excitability of postsynaptic neurons. This phenomenon was first systematically described by Terje Lømo and Per Andersen in 1973, who demonstrated that a brief, high-frequency tetanus (a rapid series of electrical stimuli) applied to the perforant path in the rabbit hippocampus resulted in a persistent enhancement of the evoked population spike in the dentate gyrus.
This discovery was groundbreaking, offering a potential cellular correlate for learning and memory, a concept that had previously been more abstract and difficult to study at a cellular level. Subsequent research has expanded upon these initial findings, elucidating the intricate molecular mechanisms and identifying its presence in various brain regions and animal models.
Primary Brain Regions Associated with LTP
While LTP can occur in many parts of the brain, it is most prominently and extensively studied in specific regions known for their roles in learning and memory. The hippocampus is arguably the most famous site, particularly its CA1 and dentate gyrus subfields, where LTP is robust and plays a critical role in spatial memory and the formation of new episodic memories.
Another key area is the amygdala, which is heavily involved in emotional learning and memory. The prefrontal cortex, essential for executive functions, working memory, and decision-making, also exhibits LTP. Furthermore, LTP has been observed in the cerebellum, crucial for motor learning and coordination, and in the visual cortex, contributing to sensory processing and adaptation. The widespread presence of LTP across these diverse brain areas underscores its fundamental importance in a broad spectrum of cognitive functions.
The Role of LTP in Learning and Memory

Alright, so we’ve laid the groundwork, understanding what Long-Term Potentiation actually is at a cellular level. Now, let’s dive into why this whole synaptic strengthening gig is so darn crucial for us to actually
- learn* things and
- remember* them. Think of it as the brain’s way of saying, “Hey, this connection is important, let’s make it stick!” It’s not just some abstract biological process; it’s the very engine driving our ability to acquire new knowledge and skills.
LTP is fundamentally about synaptic plasticity, which is the brain’s incredible ability to change its structure and function in response to experience. It’s not a static organ; it’s constantly rewiring itself. LTP is one of the most well-studied and widely accepted mechanisms underlying this plasticity, specifically demonstrating how the strength of connections between neurons can be persistently enhanced. This persistent enhancement is key to memory formation, as it allows for the encoding and consolidation of information.
When you learn something new, specific neural pathways are activated. If these pathways are repeatedly activated or activated in a significant way, LTP kicks in, making those connections stronger and more efficient, thus solidifying the memory.
LTP and New Memory Formation
The process of forming new memories is intrinsically linked to the induction and maintenance of LTP. When we encounter new information or experiences, specific patterns of neural activity are generated. If these patterns are strong enough or repeated, they trigger the molecular cascades that lead to LTP at the synapses involved. This strengthening isn’t just a temporary boost; it’s a lasting change that makes it easier for those neurons to communicate in the future.
This persistent alteration in synaptic efficacy is what allows the brain to encode and store information, transforming fleeting sensory input into stable memories.
Synaptic Plasticity as Represented by LTP
LTP is a prime example of functional synaptic plasticity. It’s a mechanism that demonstrates how the efficiency of signal transmission between neurons can be modulated over time. Unlike short-term changes, LTP results in a long-lasting increase in synaptic strength, meaning that a presynaptic neuron can evoke a stronger response in a postsynaptic neuron after the potentiation has occurred. This is achieved through a variety of cellular and molecular changes, including an increase in the number of neurotransmitter receptors on the postsynaptic membrane and changes in the presynaptic terminal’s ability to release neurotransmitters.
The enduring increase in synaptic strength, a hallmark of LTP, is the cellular basis for learning and memory.
LTP Involvement in Different Memory Types
The brain doesn’t have a single memory system; it’s a complex network with various specialized circuits. LTP plays a role across many of these, but its precise involvement can differ depending on the type of memory being formed and consolidated.
- Declarative Memory: This refers to memories that can be consciously recalled, such as facts and events (e.g., remembering your birthday, learning a historical date). The hippocampus is a critical structure for declarative memory formation, and extensive research has shown that LTP in hippocampal circuits is essential for encoding and consolidating these types of memories. When you learn a new fact, the relevant neural pathways in the hippocampus undergo LTP, making those connections more robust and accessible for later retrieval.
- Procedural Memory: This encompasses skills and habits that are learned and performed unconsciously, such as riding a bike or playing a musical instrument. While the hippocampus is less directly involved, other brain areas like the basal ganglia and cerebellum are crucial. LTP mechanisms are also implicated in these areas, contributing to the gradual strengthening of neural circuits required for motor learning and the development of ingrained habits.
The repetitive practice involved in learning a skill likely drives LTP in these procedural memory networks.
LTP Impairment and Memory Deficits
Given LTP’s fundamental role in memory, it’s no surprise that disruptions to this process are strongly linked to memory problems. When LTP mechanisms are impaired, the brain’s ability to form and retain new memories is compromised.
Numerous studies, both in animal models and in humans, have demonstrated this connection:
- Genetic Manipulations: In laboratory settings, researchers can genetically modify animals to have deficiencies in molecules crucial for LTP (like NMDA receptors). These animals often exhibit significant deficits in learning and memory tasks.
- Pharmacological Interventions: Drugs that block LTP induction or maintenance can prevent the formation of new memories in experimental subjects.
- Neurodegenerative Diseases: Conditions like Alzheimer’s disease are characterized by significant memory loss. While Alzheimer’s is a complex disease with multiple pathological processes, impaired synaptic plasticity, including compromised LTP, is considered a key contributor to the cognitive decline observed. Amyloid plaques and tau tangles, hallmarks of Alzheimer’s, are known to disrupt synaptic function and interfere with LTP.
- Aging: Even in normal aging, there can be a subtle decline in LTP capacity, which may contribute to the age-related memory difficulties experienced by some individuals. This suggests that maintaining robust LTP is important for healthy cognitive function throughout life.
Mechanisms and Models of LTP Induction

Alright, so we’ve touched upon the big picture of what Long-Term Potentiation (LTP) is and why it’s a cornerstone of how our brains learn and remember. Now, let’s dive deep into the nitty-gritty – the actual cellular dance that makes LTP happen. Think of it as understanding the intricate choreography behind that strengthened connection between neurons. It’s not magic, it’s a precise biological process, and the way it unfolds is pretty darn fascinating.This section is all about the “how.” We’ll dissect the steps of synaptic transmission that kickstart LTP, highlighting the crucial roles of specific receptor types.
We’ll also connect this to a fundamental idea in neuroscience, Hebbian learning, and then break down the different stages LTP goes through, from its immediate onset to its lasting effects. It’s like watching a skilled artisan at work, each movement deliberate and essential for the final masterpiece.
Synaptic Transmission Leading to LTP
The journey to LTP begins with a single, powerful message. When a presynaptic neuron fires an action potential, it triggers the release of neurotransmitters into the synaptic cleft, the tiny gap between neurons. These neurotransmitters then bind to receptors on the postsynaptic neuron, influencing its electrical activity. For LTP to be induced, this transmission needs to be not just any transmission, but a strong and repeated one, often described as “fire together, wire together.” This high-frequency stimulation is the initial spark that sets the stage for enduring changes in synaptic strength.
The Roles of NMDA and AMPA Receptors, What is long term potentiation in psychology
At the heart of LTP induction are two critical players on the postsynaptic membrane: NMDA receptors and AMPA receptors. These are ionotropic glutamate receptors, meaning they open channels that allow ions to pass through when glutamate binds to them. However, they have distinct properties that are key to the LTP mechanism.AMPA receptors are the primary mediators of fast excitatory neurotransmission.
When glutamate binds, they open and allow sodium ions (Na+) to rush into the postsynaptic neuron, causing a depolarization. This initial depolarization is important, but it’s not enough on its own to trigger LTP.NMDA receptors, on the other hand, are a bit more complex. Under normal resting conditions, they are blocked by a magnesium ion (Mg2+). For the NMDA receptor channel to open and allow ions like calcium (Ca2+) and sodium (Na+) to flow through, two conditions must be met simultaneously: glutamate must bind to the receptor, AND the postsynaptic membrane must be sufficiently depolarized to dislodge the Mg2+ block.
This coincidence detection mechanism is fundamental to LTP. The strong, repeated stimulation that leads to LTP causes enough depolarization through AMPA receptor activation to remove the Mg2+ block from NMDA receptors. Once unblocked, the influx of calcium ions through NMDA receptors is the crucial signal that initiates the cascade of events leading to LTP.
“When an axon of cell A is near cell B and repeatedly or persistently takes part in firing it, some growth process or metabolic change takes place in one or both cells such that A’s efficiency, as one of the cells firing B, is increased.”
Donald Hebb
Hebbian Learning and LTP
Donald Hebb’s famous postulate, often summarized as “neurons that fire together, wire together,” provides a conceptual framework for understanding LTP. In the context of LTP, Hebbian learning describes how the simultaneous activation of a presynaptic neuron and a postsynaptic neuron strengthens the synaptic connection between them. The repeated and strong firing of the presynaptic neuron, leading to the excitation of the postsynaptic neuron, is precisely the scenario that triggers the NMDA receptor-dependent calcium influx.
This calcium influx then initiates intracellular signaling pathways that lead to a long-lasting increase in the efficacy of the synapse. It’s this correlated activity that marks the synapse for strengthening, ensuring that the pathway involved in the co-activation becomes more efficient for future communication.
Phases of LTP
LTP is not a single, monolithic event; it unfolds in distinct phases, each with its own molecular mechanisms and implications for memory. Understanding these phases helps us appreciate the dynamic nature of synaptic plasticity.We can broadly categorize LTP into two main phases:
- Early-Phase LTP (E-LTP): This phase occurs relatively quickly after the induction stimulus, typically lasting for a few hours. E-LTP is primarily mediated by modifications to existing synaptic proteins. The influx of calcium through NMDA receptors triggers the activation of various protein kinases, such as CaMKII (Calcium/calmodulin-dependent protein kinase II). These kinases then lead to changes like the insertion of more AMPA receptors into the postsynaptic membrane and an increase in the conductance of existing AMPA receptors.
This results in a greater postsynaptic response to subsequent glutamate release, effectively strengthening the synapse without requiring new protein synthesis.
- Late-Phase LTP (L-LTP): This phase is characterized by its long-lasting nature, persisting for days, weeks, or even longer, and is considered crucial for long-term memory formation. L-LTP requires the synthesis of new proteins and the growth of new synaptic structures. The molecular signals initiated during E-LTP, particularly those involving sustained activation of certain signaling pathways, can lead to changes in gene expression in the postsynaptic neuron.
This results in the production of new proteins that are incorporated into the synapse, leading to more permanent structural and functional alterations. These changes can include the formation of new dendritic spines, an increase in the size of existing spines, and alterations in the number and type of receptors at the synapse, all contributing to a robust and enduring enhancement of synaptic strength.
Experimental Methods for Studying LTP: What Is Long Term Potentiation In Psychology

Investigating the intricate dance of long-term potentiation requires a sophisticated toolkit. Researchers have developed ingenious methods to observe and manipulate these synaptic changes, allowing us to peek into the very machinery of learning and memory. These techniques range from meticulously controlled laboratory settings to more naturalistic observations, each offering a unique lens through which to understand LTP.The study of LTP is fundamentally an exercise in measuring changes in synaptic strength.
This involves precisely stimulating neurons and then recording their electrical responses. By comparing these responses before and after specific interventions designed to induce LTP, scientists can quantify the magnitude and persistence of synaptic enhancement. This empirical approach forms the bedrock of our understanding of how neural connections are strengthened over time.
Electrophysiological Techniques for Measuring LTP
Electrophysiology is the cornerstone of LTP research, providing direct measurements of neuronal activity and synaptic transmission. These techniques allow for the real-time observation of changes in the electrical signals exchanged between neurons.Common electrophysiological techniques include:
- Extracellular Field Recordings: This method involves placing microelectrodes in the extracellular space near synapses. It measures the summed electrical activity of a population of neurons, often referred to as the “field potential.” A key parameter measured is the field excitatory postsynaptic potential (fEPSP), which reflects the synchronized synaptic input to a group of neurons. An increase in the fEPSP slope after a high-frequency stimulation (HFS) or other LTP-inducing stimuli is a hallmark of LTP.
- Intracellular Recordings: Here, a fine electrode is inserted directly into a neuron. This allows for the measurement of the neuron’s membrane potential, providing more detailed information about synaptic events, such as excitatory postsynaptic potentials (EPSPs) and inhibitory postsynaptic potentials (IPSPs). Intracellular recordings can reveal changes in synaptic efficacy at the single-neuron level.
- Patch Clamp Recordings: A highly refined technique where a glass pipette with a very fine tip is sealed onto a small patch of the neuronal membrane. This allows for the isolation and measurement of ion channel activity, as well as the recording of miniature postsynaptic currents (mPSCs) or potentials (mEPSPs). Changes in the frequency or amplitude of mPSCs/mEPSPs can indicate alterations in neurotransmitter release or receptor sensitivity, key components of LTP.
In Vivo and In Vitro Experimental Paradigms for LTP Research
The study of LTP can be conducted in two primary experimental settings, each offering distinct advantages and challenges. These paradigms allow researchers to investigate LTP in different levels of biological complexity.In vitro studies offer unparalleled control over experimental conditions, making them ideal for dissecting specific molecular mechanisms. In contrast, in vivo studies provide a more biologically relevant context, allowing researchers to examine LTP within the intact, functioning brain.
In Vitro Paradigms:
These experiments are typically performed on isolated brain slices or cultured neuronal networks. This approach allows for precise stimulation and recording with minimal interference from other physiological systems.
- Hippocampal Slices: The hippocampus is a brain region critically involved in learning and memory, making it a prime target for LTP research. Thin slices of hippocampus (around 300-400 micrometers thick) are prepared from rodents and maintained in a physiological solution. Stimulating electrodes are placed in the Schaffer collateral pathway and recording electrodes in the CA1 stratum radiatum to measure fEPSPs.
High-frequency stimulation (e.g., tetanic stimulation) is applied to the Schaffer collaterals to induce LTP, and the subsequent increase in fEPSP slope is monitored.
- Cultured Neuronal Networks: Neurons dissociated from brain tissue and grown in culture dishes can also be used. This allows for long-term observation of synaptic plasticity and the effects of various drugs or genetic manipulations.
In Vivo Paradigms:
These experiments are conducted on living animals, typically rodents, allowing for the study of LTP in its natural environment. While more complex, they offer crucial insights into how LTP functions within a complete neural circuit.
- Electrode Implantation: Chronic electrodes are surgically implanted into specific brain regions of awake, behaving animals. Stimulation and recording electrodes are placed to target pathways known to exhibit LTP, such as the perforant path-input to the dentate gyrus or the Schaffer collateral-CA1 synapse.
- Behavioral Correlates: In vivo studies often link LTP induction to specific behavioral tasks. For instance, LTP in the hippocampus can be induced during learning paradigms, and the strength of LTP can be correlated with the animal’s performance on memory tests.
- Optogenetics and Chemogenetics: Advanced in vivo techniques like optogenetics (using light to control genetically modified neurons) and chemogenetics (using designer drugs to control genetically modified neurons) allow for highly specific manipulation of neuronal activity and the study of their causal role in LTP and behavior.
Use of Genetic Models and Pharmacological Interventions in LTP Studies
Understanding the molecular underpinnings of LTP heavily relies on manipulating the genetic makeup and chemical environment of neurons. These approaches allow researchers to pinpoint the specific genes, proteins, and signaling pathways that are essential for LTP induction and maintenance.Genetic and pharmacological tools provide powerful means to test hypotheses about the molecular players involved in synaptic plasticity. By altering these components, scientists can observe the consequences for LTP and, by extension, for learning and memory.
- Genetic Knockout/Knock-in Models: Researchers can create animal models where specific genes involved in synaptic function are either deleted (knockout) or modified (knock-in). For example, mice lacking a gene for a particular NMDA receptor subunit might show impaired LTP. Studying these models reveals the necessity of these genes for LTP.
- Pharmacological Agents: A wide array of drugs can be used to block or enhance specific molecular targets involved in LTP. For instance, NMDA receptor antagonists (like AP5) are known to block LTP induction, providing strong evidence for the role of NMDA receptors. Conversely, drugs that enhance AMPA receptor function might facilitate LTP.
- RNA Interference (RNAi) and CRISPR-Cas9: These gene-editing technologies allow for more precise and transient knockdown or modification of gene expression in specific neurons, both in vitro and in vivo.
- Pharmacological Interventions in Behavioral Studies: Drugs affecting LTP can be administered to animals before or during learning tasks to assess their impact on memory formation and retention, thus linking molecular mechanisms to cognitive function.
Hypothetical Experimental Setup to Investigate the Effect of a Novel Compound on LTP in Hippocampal Slices
Let’s design a hypothetical experiment to explore how a novel compound, let’s call it “NeuroEnhance,” might affect LTP in hippocampal slices. This setup will utilize standard electrophysiological techniques. Objective: To determine if NeuroEnhance enhances or impairs LTP at the Schaffer collateral-CA1 synapse in rat hippocampal slices. Experimental Design:
We will employ a comparative approach, treating one group of slices with NeuroEnhance and a control group with vehicle (the solution NeuroEnhance is dissolved in) to isolate the compound’s effects.
Materials:
- Acute transverse hippocampal slices (300-400 µm thick) from adult male Sprague-Dawley rats.
- Artificial cerebrospinal fluid (aCSF) saturated with 95% O2 / 5% CO2, containing (in mM): 124 NaCl, 2.5 KCl, 1.25 NaH2PO4, 26 NaHCO3, 10 glucose, 2 CaCl2, 1 mM MgCl2.
- A perfusion system to continuously supply oxygenated aCSF to the slices at a controlled temperature (30-32°C).
- Stimulating electrode (e.g., bipolar tungsten wire) placed in the Schaffer collateral pathway.
- Recording electrode (e.g., borosilicate glass pipette filled with aCSF) placed in the CA1 stratum radiatum.
- A high-frequency stimulation (HFS) protocol to induce LTP (e.g., a train of 100 pulses at 100 Hz for 1 second, delivered once).
- A drug delivery system to perfuse slices with either aCSF containing NeuroEnhance or vehicle.
Procedure:
- Slice Preparation: Prepare hippocampal slices and transfer them to the perfusion chamber. Allow slices to equilibrate for at least 1 hour before recording.
- Baseline Recording: Record the baseline fEPSP response by delivering single pulses (at an intensity that elicits approximately 40-50% of the maximum response) every 20 seconds for 10 minutes.
- Drug Incubation: Divide the slices into two groups:
- Control Group: Perfuse with standard aCSF.
- NeuroEnhance Group: Perfuse with aCSF containing a specific concentration of NeuroEnhance (e.g., 10 µM) for 30 minutes prior to LTP induction and during the entire recording period.
- LTP Induction: After the incubation period, deliver the HFS protocol to the Schaffer collaterals in both groups.
- Post-Induction Recording: Continue recording fEPSPs at the same stimulation intensity and frequency as the baseline for at least 60 minutes after HFS.
- Data Analysis:
- Calculate the slope of the fEPSP for each recorded event.
- Normalize the fEPSP slopes to the average baseline slope for each slice.
- Compare the normalized fEPSP slopes between the control and NeuroEnhance groups during the 60-minute post-induction period.
Expected Outcomes and Interpretation:
- If NeuroEnhance enhances LTP, the fEPSP slopes in the NeuroEnhance group will show a significantly larger and more sustained potentiation compared to the control group. This would suggest that NeuroEnhance facilitates synaptic strengthening.
- If NeuroEnhance impairs LTP, the fEPSP slopes in the NeuroEnhance group will show less potentiation or even depression compared to the control group. This would indicate that NeuroEnhance interferes with synaptic strengthening.
- If NeuroEnhance has no significant effect, there will be no statistical difference in the fEPSP slopes between the two groups.
This hypothetical experiment provides a framework for rigorously testing the impact of a novel compound on a fundamental form of synaptic plasticity.
LTP and Neurological Disorders
The intricate dance of synaptic plasticity, particularly the phenomenon of Long-Term Potentiation (LTP), is fundamental to healthy brain function. When this delicate process is disrupted, it can have profound implications for cognitive abilities, leading to a spectrum of neurological disorders. Understanding how LTP goes awry in these conditions offers critical insights into their underlying mechanisms and potential avenues for therapeutic intervention.The brain’s ability to adapt and form new memories relies heavily on the strengthening of synaptic connections, a hallmark of LTP.
When this strengthening mechanism is impaired, it can manifest as difficulties in learning, memory recall, and overall cognitive decline. This widespread impact makes LTP a crucial area of investigation for a variety of neurological conditions.
Altered LTP in Alzheimer’s Disease
Alzheimer’s disease (AD) is characterized by progressive memory loss and cognitive deterioration, and research strongly suggests that impaired LTP plays a significant role in its pathogenesis. The accumulation of amyloid-beta plaques and tau tangles, the pathological hallmarks of AD, are known to interfere with the molecular pathways essential for LTP induction and maintenance. Specifically, these pathological proteins can disrupt the function of NMDA receptors and AMPA receptors, key players in the synaptic changes that underlie LTP.
This disruption leads to a reduced ability of synapses to strengthen, directly impacting the formation and retrieval of memories.
The synaptic dysfunction observed in Alzheimer’s disease is often characterized by a deficit in long-term potentiation, leading to impaired memory formation and consolidation.
Studies have shown that in animal models of AD, there is a marked reduction in LTP in hippocampal regions, which are critical for memory. This deficit correlates with behavioral impairments in learning and memory tasks. Furthermore, therapeutic strategies aimed at clearing amyloid-beta or reducing tau pathology have shown some promise in restoring LTP and improving cognitive function in these models, underscoring the direct link between LTP integrity and AD progression.
LTP Dysfunction in Other Neurodegenerative Disorders
The implications of LTP dysfunction extend beyond Alzheimer’s disease to encompass a range of other neurodegenerative disorders. In Parkinson’s disease, while primarily known for motor deficits, cognitive impairments, including memory problems, are also common. Research indicates that dopamine depletion, a core feature of Parkinson’s, can also negatively impact synaptic plasticity mechanisms like LTP, particularly in areas like the striatum and hippocampus.
This disruption contributes to the cognitive deficits experienced by some Parkinson’s patients.Similarly, in Huntington’s disease, a genetic disorder characterized by progressive motor, cognitive, and psychiatric symptoms, LTP has been found to be impaired in various brain regions. The mutant huntingtin protein is thought to interfere with synaptic function and plasticity, leading to deficits in learning and memory.Epilepsy, characterized by recurrent seizures, also involves aberrant synaptic plasticity.
While some forms of plasticity might be enhanced in epilepsy, leading to hyperexcitability, the capacity for normal, adaptive LTP can be compromised, affecting learning and memory in individuals with the condition.
Research on LTP Informing Therapeutic Strategies
The profound link between LTP and neurological disorders opens exciting avenues for developing novel therapeutic strategies. By understanding the specific molecular mechanisms that are disrupted in conditions like AD, researchers can target these pathways to restore or enhance synaptic plasticity.For example, drug development efforts are focused on:
- Modulating NMDA and AMPA receptor function to improve synaptic transmission and LTP.
- Developing agents that can clear pathological proteins like amyloid-beta and tau, thereby alleviating their inhibitory effects on LTP.
- Investigating compounds that can promote neurotrophic factors, such as BDNF (Brain-Derived Neurotrophic Factor), which are known to support synaptic plasticity and LTP.
- Exploring non-invasive brain stimulation techniques, like transcranial magnetic stimulation (TMS) or transcranial direct current stimulation (tDCS), which have shown potential in modulating synaptic plasticity and improving cognitive function in certain neurological conditions.
The success of these therapeutic approaches hinges on precisely identifying which aspects of LTP are compromised in a given disorder and then developing interventions that can selectively restore those functions without causing adverse effects.
Potential Biomarkers Related to LTP
The development of reliable biomarkers is crucial for early diagnosis, monitoring disease progression, and assessing treatment efficacy in neurological disorders. Given its central role in cognitive function, LTP-related markers hold significant promise.Potential biomarkers related to LTP could include:
- Molecular Markers: Measuring levels of key proteins involved in LTP, such as phosphorylated NMDA receptors, AMPA receptor subunits (e.g., GluR1), or downstream signaling molecules like CaMKII, in cerebrospinal fluid (CSF) or blood.
- Genetic Markers: Identifying genetic variations that influence LTP efficiency or susceptibility to LTP-related cognitive decline.
- Electrophysiological Markers: Using techniques like electroencephalography (EEG) or magnetoencephalography (MEG) to assess synaptic function and plasticity in response to specific stimuli. For instance, changes in evoked potentials or oscillatory activity patterns could reflect altered LTP.
- Neuroimaging Markers: Advanced neuroimaging techniques, such as functional magnetic resonance imaging (fMRI) combined with specific cognitive tasks designed to probe memory encoding and retrieval, could reveal functional deficits in brain regions critical for LTP. Positron emission tomography (PET) tracers that bind to synaptic proteins or receptors involved in LTP could also serve as valuable biomarkers.
The identification and validation of these LTP-related biomarkers would represent a significant advancement in the clinical management of neurological disorders, allowing for more personalized and effective treatments.
Factors Influencing LTP

Long-Term Potentiation isn’t a monolithic event; it’s a dynamic process heavily influenced by a symphony of factors, from the chemical messengers zipping around to the overall state of our being. Understanding these influences helps us grasp why LTP can be so robust and adaptable, or sometimes, frustratingly elusive. It’s like tuning a finely-tuned instrument – small adjustments can make a big difference in the final melody of memory formation.Beyond the primary players like glutamate, a whole cast of other neurochemicals and physiological conditions can either boost or dampen the LTP process.
These interactions are crucial for fine-tuning synaptic strength and ensuring that only the most relevant information gets etched into our neural circuitry for the long haul.
Neurotransmitter Modulation of LTP
While glutamate is the star of the LTP show, other neurotransmitters play significant supporting roles, acting as crucial modulators. These can either enhance or inhibit the induction and maintenance of LTP, depending on their type, concentration, and the specific receptors they bind to.
- GABA (Gamma-Aminobutyric Acid): As the primary inhibitory neurotransmitter, GABA generally acts to suppress neuronal activity. In the context of LTP, GABAergic signaling can influence the threshold for LTP induction. For instance, by reducing overall excitability, GABA can prevent the uncontrolled firing that might lead to aberrant plasticity. However, under certain conditions, disinhibition mediated by GABA can actually facilitate LTP by allowing for more precise timing of excitatory inputs.
- Serotonin: This neuromodulator, involved in mood and cognition, can also impact LTP. Serotonergic pathways can influence the excitability of neurons and the release of other neurotransmitters, thereby indirectly affecting LTP. Research suggests that serotonin can, in some brain regions and under specific experimental paradigms, enhance LTP by acting on specific serotonin receptor subtypes.
- Norepinephrine: Similar to serotonin, norepinephrine is involved in attention, arousal, and stress responses. Its influence on LTP is complex and context-dependent. In some cases, norepinephrine can promote LTP, particularly when it’s associated with salient or arousing events, aligning with its role in memory consolidation during wakefulness.
Neuromodulator Impact on LTP
Neuromodulators like dopamine and acetylcholine are critical for orchestrating complex brain functions, including learning and memory, and they exert their influence on LTP through diverse mechanisms. Their presence can significantly alter the plasticity landscape.
- Dopamine: Dopamine is famously linked to reward, motivation, and motor control, but it also plays a vital role in synaptic plasticity. Dopaminergic signaling can facilitate LTP induction, especially in pathways associated with reward learning. It can enhance the probability of glutamate release and influence the sensitivity of NMDA receptors, making it easier for synapses to strengthen. For example, studies in the striatum, a region rich in dopamine, show that dopamine can be essential for certain forms of LTP related to habit formation.
- Acetylcholine: This neurotransmitter is crucial for attention, arousal, and memory, particularly in the hippocampus. Acetylcholine can enhance LTP induction by acting on muscarinic and nicotinic receptors on both pre- and post-synaptic neurons. It can increase neuronal excitability, modulate glutamate release, and even influence the downstream signaling cascades involved in plasticity. The enhanced learning observed during states of focused attention is often attributed, in part, to acetylcholine’s facilitatory effects on LTP.
Influence of Physiological States on LTP
Our internal environment, shaped by daily rhythms and emotional states, profoundly impacts our brain’s ability to form and retain memories. These physiological conditions can alter the neurochemical milieu and neuronal excitability, thereby influencing LTP.
- Sleep: Sleep is not merely a period of rest; it’s an active state crucial for memory consolidation. During sleep, particularly slow-wave sleep, there is evidence suggesting a form of synaptic downscaling, which paradoxically helps to consolidate important memories by weakening less relevant ones. Furthermore, certain sleep stages may be conducive to the stabilization of LTP that was induced during wakefulness.
Disruptions in sleep patterns are often associated with impaired learning and memory, underscoring the importance of sleep for effective LTP.
- Stress: The impact of stress on LTP is a double-edged sword. Acute, moderate stress can sometimes enhance LTP and memory formation, likely by releasing stress hormones like cortisol and norepinephrine, which can prime the brain for learning. However, chronic or severe stress can be detrimental. Prolonged exposure to stress hormones can impair LTP induction and maintenance, particularly in the hippocampus, leading to deficits in learning and memory.
This is often observed in conditions like post-traumatic stress disorder (PTSD).
Effects of Different Stimulation Patterns on LTP Magnitude and Duration
The way we stimulate neurons to induce LTP has a direct bearing on how strong and how long that potentiation lasts. It’s not just about
- if* we stimulate, but
- how* we stimulate. Different patterns of electrical or chemical input can lead to distinct outcomes in synaptic strength.
High-Frequency Stimulation (HFS) vs. Theta-Burst Stimulation (TBS)
High-frequency stimulation (HFS), typically involving trains of stimuli at 100 Hz or higher, is a classic method for inducing robust LTP. This intense burst of activity leads to a rapid influx of calcium into the postsynaptic neuron, activating downstream kinases essential for LTP. However, HFS can sometimes lead to saturation of plasticity or even excitotoxicity if not carefully controlled.
Theta-burst stimulation (TBS), which mimics the natural theta rhythm (4-8 Hz) observed in the hippocampus during learning and memory tasks, is often considered a more physiologically relevant pattern for inducing LTP. TBS can be delivered in either a high-frequency burst (e.g., 4-5 pulses at 100 Hz repeated at 5 Hz) or a low-frequency burst. TBS is generally effective at inducing long-lasting LTP with a lower risk of excitotoxicity compared to HFS.
The magnitude and duration of LTP induced by TBS can vary depending on the specific parameters of the burst and the frequency of repetition.
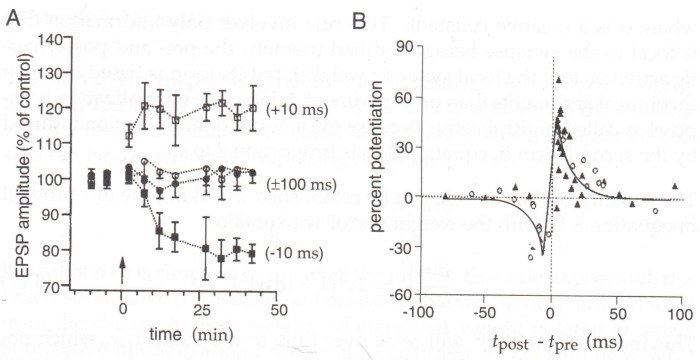
Paired-Pulse Stimulation (PPS) and Spike-Timing-Dependent Plasticity (STDP)
Paired-pulse stimulation (PPS) involves delivering two pulses of stimulation with a short interval between them. The response to the second pulse, relative to the first, can reveal information about presynaptic release probability and short-term plasticity. While not a direct method for inducing LTP, the interval in PPS can influence subsequent plasticity. For instance, short intervals (e.g., 20-50 ms) can lead to paired-pulse facilitation, which might prime a synapse for LTP induction.
Spike-timing-dependent plasticity (STDP) is a more sophisticated form of plasticity where the precise timing of pre- and postsynaptic action potentials determines whether LTP or LTD (Long-Term Depression) occurs. If the presynaptic neuron fires just before the postsynaptic neuron, LTP is typically induced (a “Hebbian” learning rule). If the presynaptic neuron fires after the postsynaptic neuron, LTD is induced. The time window for STDP is usually very narrow, on the order of milliseconds.
The magnitude and duration of plasticity induced by STDP are highly dependent on the precise timing difference between the spikes, with larger timing differences often leading to more robust and enduring changes in synaptic strength.
LTP in Different Neural Circuits

Alright, so we’ve been deep-diving into the nitty-gritty of Long-Term Potentiation, mainly focusing on its star performance in the hippocampus. But hey, the brain’s a vast place, and LTP isn’t some one-trick pony confined to memory central. It’s actually showing up in a bunch of other brain regions, each with its own special gig, shaping how we learn and react in ways you might not even realize.Think of LTP as a universal language of neural plasticity, spoken with slightly different dialects across the brain.
While the core mechanism of strengthening synaptic connections remains, the specific players, the timing, and the ultimate outcome can vary quite a bit depending on where it’s happening. This adaptability is what allows our brains to handle such a diverse range of tasks, from remembering a scary encounter to mastering a new dance move.
LTP in Non-Hippocampal Brain Areas
While the hippocampus gets a lot of the spotlight for its role in forming new memories, LTP is far from being exclusive to this region. Evidence points to its significant presence and function in other key brain areas, each contributing to distinct cognitive and behavioral processes. This widespread distribution underscores LTP’s fundamental importance as a mechanism for neural plasticity throughout the brain.LTP has been extensively documented in areas such as the cerebral cortex, the amygdala, and the cerebellum.
In the cortex, it’s implicated in perceptual learning, where repeated exposure to stimuli can lead to enhanced recognition and discrimination. For instance, learning to distinguish subtle differences in visual patterns or auditory tones often involves cortical LTP. The amygdala, crucial for processing emotions, utilizes LTP for the formation of emotional memories, particularly those associated with fear. The cerebellum, traditionally known for motor control, also exhibits LTP, which is vital for motor learning and skill acquisition.
These examples highlight that LTP is a pervasive phenomenon, adapting its role to the specific functional demands of different neural circuits.
Functional Significance of LTP in the Amygdala for Emotional Learning
The amygdala, often described as the brain’s “fear center,” is a critical hub for processing emotions, especially fear and threat detection. Long-term potentiation plays a pivotal role here, acting as the cellular substrate for how we learn and remember emotionally charged events. When you have a scary experience, like encountering a dog that barks aggressively, the neural pathways in your amygdala that associate that specific dog (or even similar-looking dogs) with danger become strengthened through LTP.This potentiation means that future encounters with similar stimuli will trigger a stronger and faster fear response.
It’s this mechanism that allows for rapid learning of what to avoid. For instance, if you were startled by a loud noise in a particular room, LTP in the amygdala might contribute to developing an aversion to that room, even if the noise was a one-off event. This is crucial for survival, as it helps us quickly adapt our behavior to avoid potential harm.
The strength and persistence of these LTP-induced changes are directly linked to the intensity and emotional valence of the original experience.
How LTP in the Cerebellum Contributes to Motor Learning
The cerebellum, a structure nestled at the back of our brain, is the undisputed maestro of motor control and coordination. It’s not just about executing movements; it’s about refining them, making them smooth, precise, and eventually, automatic. LTP in the cerebellum is the silent architect behind this incredible ability to learn and master motor skills, from riding a bike to playing a musical instrument.When you practice a new motor skill, like hitting a tennis ball, your cerebellum is constantly receiving feedback about your movements.
LTP occurs at specific synapses within the cerebellar circuitry, particularly involving the climbing fibers and Purkinje cells. These synapses are strengthened based on the accuracy of your movements. If your swing is effective, the relevant neural connections are potentiated, making that successful motor pattern more likely to be repeated. Conversely, if your movement is inaccurate, inhibitory signals can lead to long-term depression (LTD), weakening those less effective pathways.
Over time, this dynamic interplay of LTP and LTD sculpts the neural networks, leading to the refined, efficient motor programs that we recognize as learned skills.
Variations in LTP Mechanisms Across Different Neuronal Populations
It’s not a one-size-fits-all situation when it comes to LTP. While the fundamental principle of synaptic strengthening is conserved, the specific molecular machinery and the precise triggers for LTP can differ significantly across various neuronal populations and brain regions. These variations allow LTP to be finely tuned to the unique demands of each circuit.For example, in the hippocampus, NMDA receptor activation is a classic requirement for LTP induction.
However, in other areas, like the cerebellum, while NMDA receptors are present, other mechanisms involving metabotropic glutamate receptors or even distinct forms of synaptic plasticity might play a more prominent role in certain pathways. The types of ionotropic glutamate receptors involved, the downstream signaling cascades activated, and the specific protein kinases that mediate the potentiation can all show regional variations.
- Hippocampus: Primarily relies on NMDA receptor-dependent mechanisms for induction, leading to widespread changes in AMPA receptor trafficking and function.
- Amygdala: Exhibits LTP that is often modulated by neuromodulators like norepinephrine and dopamine, enhancing the consolidation of emotionally salient memories.
- Cerebellum: Characterized by LTP at specific synapses, such as those involving climbing fibers onto Purkinje cells, which is crucial for error-driven motor learning.
- Cerebral Cortex: LTP in cortical areas can be more complex, involving interactions between different receptor types and contributing to synaptic plasticity underlying sensory and cognitive learning.
These variations highlight the remarkable adaptability of neural systems. The same basic principle of strengthening connections is employed, but the specific “how” and “when” are exquisitely tailored to the functional role of each brain region, enabling a diverse array of learning and memory processes.
Computational Models of LTP

Alright, so we’ve delved deep into the nuts and bolts of Long-Term Potentiation, from its foundational underpinnings to its role in memory and even its connection to neurological woes. Now, let’s shift gears and talk about how we can actuallysimulate* this whole synaptic plasticity jazz on a computer. It’s not just about watching neurons fire; it’s about building digital brains to understand the biological ones.
Computational models are our virtual petri dishes, allowing us to tweak parameters and see what happens without zapping any actual rats or people.Computational models of LTP are essentially mathematical frameworks designed to replicate the complex biophysical and biochemical processes that underlie synaptic strengthening. They take the known biological mechanisms of LTP and translate them into equations and algorithms. Think of it as building a sophisticated Lego set of a synapse, where each brick represents a protein, a ion channel, or a signaling cascade, and the rules of assembly are the biological laws governing LTP.
These models are not just theoretical exercises; they are powerful tools for testing hypotheses, exploring scenarios that are difficult or impossible to study experimentally, and ultimately, for gaining a more profound understanding of how memories are formed and stored at the neural level.
Principles Behind Computational Models Simulating LTP
The core principle behind computational models of LTP is to capture the essence of synaptic plasticity by representing the key biological players and their interactions. This involves translating molecular events into mathematical relationships. These models often focus on the influx of calcium ions through NMDA receptors, which is a critical trigger for LTP, and the subsequent cascade of intracellular signaling pathways that lead to changes in AMPA receptor trafficking and function.
The models aim to reproduce the input-output relationship of a synapse, showing how a specific pattern of stimulation leads to a lasting increase in synaptic efficacy.At their heart, these models are governed by principles of dynamic systems and signal transduction. They often employ differential equations to describe the rate of change of various molecular concentrations or the state of ion channels over time.
Key components that are typically modeled include:
- Receptor Dynamics: Simulating the binding of neurotransmitters to postsynaptic receptors like NMDA and AMPA, and how their conductance changes based on voltage and ligand availability.
- Calcium Influx and Buffering: Modeling the entry of calcium ions into the postsynaptic neuron and how intracellular calcium levels are regulated by buffering proteins and pumps.
- Kinase and Phosphatase Activity: Representing the activation and deactivation of key enzymes like CaMKII and protein phosphatases, which play crucial roles in downstream signaling.
- Receptor Trafficking: Simulating the insertion and removal of AMPA receptors from the postsynaptic membrane, a major mechanism for altering synaptic strength.
- Gene Expression and Protein Synthesis: In more complex models, these can be included to account for long-lasting forms of LTP.
These elements are interconnected, forming a network of interactions that, when subjected to specific input patterns (mimicking high-frequency stimulation or correlated activity), result in a sustained potentiation of the synaptic response.
How Computational Models Help Researchers Understand Synaptic Plasticity
Computational models act as virtual laboratories, allowing researchers to conduct experiments that would be prohibitively complex or impossible in biological systems. By manipulating parameters within the model, scientists can isolate the effects of specific molecular players or signaling pathways on LTP induction and maintenance. This helps to disentangle the intricate web of interactions that occur within a synapse. For instance, a model can be used to test the hypothesis that a particular kinase is essential for LTP by artificially reducing its activity in the simulation and observing the outcome.These models are invaluable for:
- Hypothesis Testing: Providing a rigorous framework to test specific hypotheses about the mechanisms of LTP. If a model based on a certain hypothesis fails to reproduce experimental data, it suggests the hypothesis needs refinement.
- Parameter Sensitivity Analysis: Identifying which parameters are most critical for LTP induction and maintenance. This can guide experimental efforts towards the most promising targets.
- Exploring Non-Linear Dynamics: Synaptic plasticity often involves non-linear processes. Models can reveal complex emergent behaviors that might not be intuitive from studying individual components in isolation.
- Bridging Scales: Connecting molecular-level events to network-level phenomena. By integrating models of single synapses with models of neural circuits, researchers can explore how synaptic plasticity contributes to learning and behavior.
- Predicting Experimental Outcomes: Suggesting novel experiments or predicting the results of existing ones, thus accelerating the pace of discovery.
The ability to systematically explore the parameter space of LTP mechanisms allows for a deeper, more quantitative understanding of synaptic plasticity than might be achievable through empirical studies alone.
Predictive Power of Computational Models for LTP-Related Phenomena
The true power of computational models lies in their ability to predict phenomena that can then be tested experimentally. These predictions can range from the specific timing of molecular events during LTP induction to the functional consequences of altering synaptic plasticity on learning and memory tasks. For example, models have been used to predict the optimal stimulation patterns required to induce LTP, the dose-response curves for drugs that modulate synaptic plasticity, and the impact of genetic mutations on learning abilities.A notable example of this predictive power is seen in models that forecast the effects of neuromodulators.
By incorporating the effects of neurotransmitters like dopamine or acetylcholine into LTP models, researchers can predict how these neuromodulators might enhance or impair learning and memory consolidation. For instance, a model might predict that a certain level of dopamine release during learning will lead to a more robust and lasting potentiation of specific synapses, which could then be experimentally verified.Another area where predictive power is crucial is in understanding neurological disorders.
Models of LTP can be modified to incorporate known deficits associated with conditions like Alzheimer’s disease or schizophrenia.
By simulating these altered states, researchers can predict how synaptic dysfunction might manifest in terms of learning deficits or cognitive impairments, guiding the search for therapeutic interventions.
For example, if a model incorporating reduced NMDA receptor function (as seen in some models of Alzheimer’s) fails to induce LTP under standard conditions, it predicts that a pharmacological intervention aimed at boosting NMDA receptor activity might be beneficial. These predictions, when validated by experimental data, significantly advance our understanding and potential treatment of these complex conditions.
Schematic Representation of a Computational Model for LTP
Let’s sketch out a simplified schematic of a computational model for LTP, focusing on the key players and their interactions. Imagine this as a simplified diagram of a postsynaptic dendritic spine.
Long-term potentiation in psychology, a crucial mechanism for learning and memory, hinges on how neural connections strengthen over time. Understanding this process is vital when considering that humans and animals are, fundamentally, are subjects of psychological research , providing the very foundation for studying how long-term potentiation shapes our cognitive abilities.
| Presynaptic Terminal | ||
| Neurotransmitter Release | ↓ | Synaptic Cleft |
| Postsynaptic Dendritic Spine | ||
| Receptors | NMDA Receptor | ↔ Voltage & Ca2+ Influx |
| AMPA Receptor | ↔ Neurotransmitter Binding & Conductance | |
| (Inferred: Number of AMPA Receptors) | ↑↓ Trafficking | |
| Intracellular Signaling | [Ca2+]i | ↑↓ Influx, Buffering, Pumps |
| CaMKII (Active/Inactive) | ↔ [Ca2+]i & Autophosphorylation | |
| Other Kinases/Phosphatases | ↔ [Ca2+]i & Signaling Cascades | |
| Phosphorylation of AMPA Receptors | ↓ CaMKII & Others | |
| Synaptic Strength (Efficacy) | ↑↓ Based on AMPA Receptor Number & Phosphorylation | |
- Stimulation Intensity/Frequency: This is the primary input parameter, mimicking the pattern of neural activity. Higher intensity or frequency is typically required to open the NMDA receptor channel and allow significant calcium influx.
- NMDA Receptor Open Probability: A function of membrane voltage and glutamate binding. This parameter dictates how much calcium enters.
- Intracellular Calcium Concentration ([Ca2+] i): A dynamic variable influenced by influx, buffering, and extrusion. It’s a critical signal.
- CaMKII Activation State: A key enzyme whose activation is dependent on [Ca 2+] i and its own autophosphorylation. Its level of activity directly influences downstream events.
- AMPA Receptor Phosphorylation State: Phosphorylation by CaMKII and other kinases can increase AMPA receptor conductance and stability.
- Number of Surface AMPA Receptors: This is a crucial outcome variable. It is modulated by insertion (requiring CaMKII activity and other factors) and removal.
- Synaptic Efficacy (Output): The ultimate measure of LTP, often represented by the postsynaptic potential amplitude. This is a direct consequence of the number and phosphorylation state of surface AMPA receptors.
In a computational model, each of these elements would be represented by variables and equations. For example, the change in [Ca 2+] i over time might be described by a differential equation incorporating terms for calcium influx through NMDA receptors, diffusion, buffering, and active pumping out of the spine. The model would then simulate how a specific pattern of presynaptic stimulation leads to changes in these variables, ultimately resulting in a sustained increase in synaptic efficacy, thus simulating LTP.
Wrap-Up
In essence, understanding what is long term potentiation in psychology reveals the remarkable plasticity of our brains. It’s a testament to the intricate biological machinery that enables us to acquire new knowledge, form lasting memories, and adapt to our environment. The ongoing research into LTP not only deepens our comprehension of normal cognitive function but also illuminates potential pathways for addressing neurological disorders and enhancing learning capabilities, painting a vivid picture of a brain in constant, purposeful flux.
Essential FAQs
What is the basic idea behind LTP?
The fundamental concept is that “neurons that fire together, wire together.” When two neurons repeatedly activate each other, the connection between them becomes stronger, making it easier for them to communicate in the future.
Where in the brain is LTP most studied?
The hippocampus is a primary region where LTP is extensively studied due to its critical role in forming new memories. However, LTP is also observed in other brain areas like the amygdala and the cortex.
How is LTP different from short-term memory?
LTP is the cellular basis for long-term memory, meaning the changes in synaptic strength are enduring. Short-term memory, on the other hand, involves more transient changes in neuronal activity that don’t necessarily lead to lasting structural modifications.
Can LTP be disrupted?
Yes, LTP can be disrupted by various factors, including certain drugs, genetic mutations, and conditions that impair brain function, leading to memory deficits.
Does LTP only involve strengthening connections?
While LTP specifically refers to the strengthening of synaptic connections, the opposite process, long-term depression (LTD), involves the weakening of these connections, which is also crucial for learning and memory.




